About Us
A Different Brand of Experts
Cambrex is a leading global contract development and manufacturing organization (CDMO) that provides drug substance development and manufacturing across the entire drug lifecycle, as well as comprehensive analytical services. Our people set us apart. We’re passionate about what we do, and this translates into an expert team that you’ll enjoy working with. We’re different because our approach is different. As creative problem-solvers, we work closely with our customers and share your drive to advance your therapies to market.


Leadership
At Cambrex, our people add a dimension that elevates our science to a new level. Everything begins with our leaders, who call upon their extensive life science experience to motivate and inspire our employees. They guide our growing team of scientific experts dedicated to the success of your programs.
Learn More
Mission and Values
We enable our customers to develop and deliver therapeutic solutions that improve the quality of life for patients around the world. We foster an engaging workplace and collaborate closely with our customers to help them navigate drug development challenges.
Learn More
Awards
We’re proud to have received recognition in the industry for delivering exceptional customer experiences and providing superior technical expertise, all while continuing to maintain the highest level of quality.
Learn More
History
With more than 40 years of experience, we have grown and expanded our global development and manufacturing network by investing in robust technology and quality infrastructure. Over time, we have established an exemplary quality and regulatory track record.
Learn More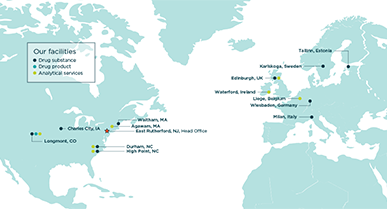
Locations
Cambrex maintains a global presence with 14 locations across North America and Europe and a corporate headquarters in East Rutherford, NJ, USA.
Learn More
Recruitment
Our people set us apart. Although our team has more than 2,000 members — distributed around the world — our workplace culture fosters close relationships and friendships. We are driven to work together in the interest of achieving shared goals.
Learn MoreWork With Us
When you work with Cambrex, you will experience a different kind of CDMO — collaborative, connected, and committed to your success. Here are five reasons you can count on Cambrex:
- We specialize in end-to-end drug substance, drug product, and analytical services across the entire drug lifecycle.
- We deliver quality in every aspect of our work, across all facilities, systems, and teams.
- Our strong commitment to the environment, health, and safety underpins everything we do.
- Our people add value to your end goals and help grow your business.
- Our global presence and local experts deliver excellence at 15 locations around the world.
Work For Us
Create a better world at Cambrex. If you’re looking for a dynamic career move, look no further. We are a team of experts, creative problem-solvers, and collaborators. We are passionate about delivering life-changing therapies to patients. We offer competitive salaries and benefits packages along with opportunities for professional development.
Check out our vacancies and consider joining our team of experts who enjoy what they do.
Sustainability at Cambrex
We are fully committed to operating ethically, protecting our workforce and the environment, and enhancing our supply chain practices.
Operate Ethically
It’s not just what we do. It’s how we do it. We operate our business in an open and ethical manner.
Protect Our Workforce
We are fully committed to ensuring the health and safety of our employees. We follow a safety-first policy and always operate proactively to manage risk and avoid accidents and injuries.
Protect the Environment
Our commitment to protecting the environment is reflected in the programs in place at each Cambrex facility to reduce emissions and institute environmentally conscious initiatives. We promote strict environmental compliance everywhere we operate, and we’re fully committed to continuously improving our performance on environmental benchmarks.
Enhance Our Supply Chain
We have a Supplier Code of Conduct in place to ensure that our suppliers use socially responsible practices that reflect our commitment to environmental, social, and governmental goals.





